Sedimentary gochemical characteristics of the Redox-sensitive elements in Ross Sea, Antarctica: Implications for paleoceanography
-
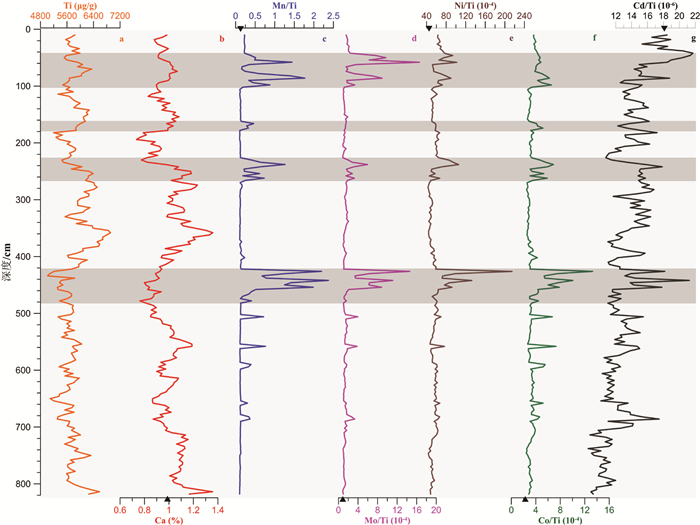
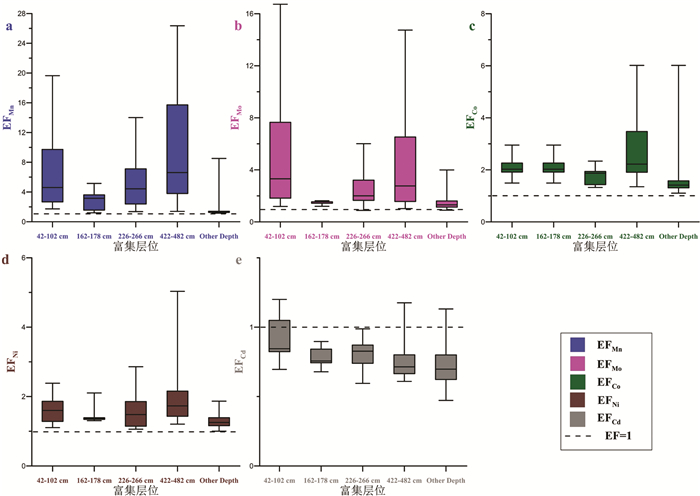
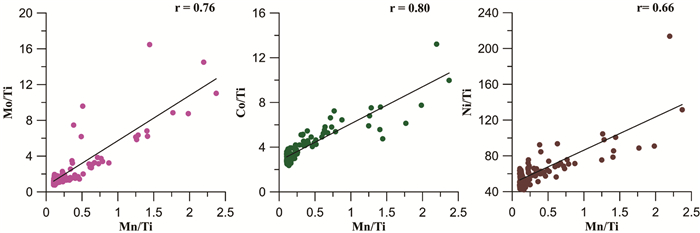
摘要: 大洋深部氧化还原环境与深部水体流通状况以及表层水体生产力密切相关。表层生产力与深部流通性变化影响着有机碳-呼吸CO2的转化及其在海洋-大气中的转移,最终与大气CO2分压(pCO2)变化密切相关。故探明大洋深部氧化还原环境的变化对于解决大气pCO2冰期旋回机制具有重要意义。本次研究以中国第31和32次南极科考获得的南极罗斯海柱状岩心ANT31-R23及表层样为研究材料。通过元素钙、钛,以及氧化还原敏感元素(RSE)锰、钼、镍、钴、镉的测试分析,以表层样中RSE与Ti的比值作为判断ANT31-R23孔中相应RSE富集程度的背景值。结果显示,Mn在沉积期均表现出富集,表明罗斯海深部在该孔沉积期为氧化环境。根据Mn在不同层位出现的富集峰识别出4次强氧化脉冲事件,可能由南大洋底层水流通性增强和/或生产力降低导致。4次氧化脉冲事件层位中Mo、Ni、Co的明显富集,是由于锰(氢)氧化物对其捕获或吸附所致。此外,推测分析认为罗斯海对冰期大气pCO2降低似乎没有明显贡献,但很可能对冰消期大气pCO2迅速升高起重要作用。然而这些有关南极罗斯海深部氧化还原环境与大气pCO2变化之间关联的推测,有待后续该孔精确年代模式的构建,方可进一步验证。Abstract: Redox conditions of deep ocean are supposed closely related to deep ocean circulation and surface water production. Facts prove that surface water production and deep water circulation may strongly influence the formation of respiration carbon and its migration from ocean interior to atmosphere, which is closely related to the rise of atmospheric pCO2. Hence, verifying the redox environment evolution of the ocean could help us clarify the mechanism of variation in atmospheric pCO2 in glacial-interglacial cycles. Samples from core ANT31-R23 and the surface sediment of central Ross Sea, which were taken by R/V Xuelong in the 31st and 32th Chinese National Antarctic Research Expedition, are used as research materials in this study. Both the major and minor elements are analyzed, including calcium, titanium and the elements sensitive to paleo-redox environment of deposition, so-called Redox-sensitive elements (RSE), such as manganese, molybdenum, nickel, cobalt and cadmium. RSEs normalized by Ti are adopted as background values to estimate if the RSEs are enriched or depleted. The result shows that enrichment of Mn occurs in the entire core indicating an oxidizing condition. Four strong oxidation pulse events are identified based on Mn peaks in different depths, which may be related to stronger circulation conditions and/or lower surface water production in the Southern Ocean during late Quaternary. The layers enriched by Mo, Co and Ni in addition to Mn, are resulted from absorption, capture or scavenge by Mn-oxyhydroxides. These results suggest that the Ross Sea does not have significant contribution to the reducing of atmospheric pCO2 during glaciation. The strong oxidation pulse events, however, may play an important role in elevating atmospheric pCO2 during deglaciation. Nevertheless, the detailed processes of this mechanism will be effectively revealed by follow-up work after the establishment of accurate chronology framework.
-
Keywords:
- Ross Sea /
- Redox-sensitive element /
- oxygenation pulse /
- ventilation /
- carbon cycle
-
蛇纹石化常指中低温环境下,橄榄石和辉石等与水相互作用,发生蚀变形成蛇纹石的过程,同时形成水镁石、氢气和磁铁矿等,并释放热量[1],在含碳体系中还会产生费托反应,由氢与碳在磁铁矿催化下形成甲烷为主的短链烷烃和其他有机化合物[2-4]。蛇纹石化伴生的无机成因烷烃气体可聚集成藏[5-6],可在海底滋养无光合作用的化能自养生物群落,通过无机过程为生命的形成与演化提供物质和能量[2,7-9]。因此,蛇纹石化研究在能源资源和地球生命起源方面均具有重要的科学价值,是近年来地球科学领域的研究热点[4,7,10-11]。
马里亚纳弧前蛇纹岩泥火山是俯冲带弧前地幔楔发生大规模蛇纹石化的结果,它由泥质、粉砂质蛇纹石胶结蛇纹岩或蛇纹石化超基性岩及变质岩等岩屑或岩块组成,具有碎屑沉积结构[7,10]。20世纪80年代中期通过地球物理、海山形态学和沉积蛇纹岩的岩石学研究,Fryer等首次提出马里亚纳弧前的串珠状海山为泥火山成因,认为它们是地幔楔大规模发生蛇纹石化的产物[12-13]。随着板块俯冲深度的不断增加,洋壳发生压实、脱水作用,产生流体与上盘地幔橄榄岩发生蛇纹石化,导致密度低、塑性强的蛇纹岩泥浆在压力与浮力的驱动下沿弧前深断裂向上运移,同时携带破碎的岩块至海底,形成具有沉积结构的蛇纹岩海山[1,7,10,14]。
马里亚纳俯冲带弧前海山是目前已知的全球唯一正在活动的蛇纹岩泥火山群,它们直径为10~30 km、高度为0.5~2 km,在距离海沟轴线30~100 km的弧前呈串珠状分布[7,10]。蛇纹岩泥火山在地质历史时期也有发育,目前在全球汇聚边缘的弧前地层和混杂岩中已发现三十多处具有沉积结构的蛇纹岩,其中十多处被认为很可能是泥火山成因[15-20]。蛇纹岩泥火山作为联系海底与岩石圈深部的纽带,其发育的岩石及流体均携带地球深部信息,被认为是认识板块俯冲过程的最佳窗口[7,14,21-22]。
蛇纹岩泥火山大规模的泥浆喷发活动停止或者间歇期间,海山顶部及裂缝带仍可发育富含氢气和无机成因甲烷的低温(约2 ℃)、强碱性(pH值高达12.5)的流体渗漏活动,对确认蛇纹岩海山的泥火山成因至关重要[23-30]。这些流体渗漏活动常伴生自生沉积,以烟囱状为主,少量呈胶结物产出[7,10,31]。1978年 “阿尔文”号深潜器在马里亚纳弧前Conical海山顶发现了由文石、方解石构成的碳酸盐岩烟囱[32],随后在其他蛇纹岩泥火山也陆续发现类似的烟囱状自生沉积,主要有水镁石质烟囱和碳酸盐质烟囱[7,28]。距离海沟较近的蛇纹岩泥火山发育水镁石质烟囱,距离海沟较远的蛇纹岩泥火山发育碳酸盐质烟囱,这种自生沉积类型与海沟距离显示系统性差异的特征,与相应泥火山渗漏流体性质相对应[7,27,29,33-34]。因此,烟囱状自生沉积记录了渗漏流体特征和俯冲带深部地球化学过程,为认识渗漏流体活动提供了可靠线索。
水镁石烟囱成因简单,为高pH值、低碳酸盐碱度流体渗漏至海底,与富镁海水混合形成的类似泉华沉积,由于水镁石不稳定,在海水中易发生溶解,导致水镁石质烟囱体型细小,一般仅数厘米到数十厘米高[7,35]。而碳酸盐岩烟囱通常体型较大,甚至可达数米高的建造[7, 30],且矿物组成多样,可能包括方解石、文石和无定形相等[36],记录了复杂的流体信息及成岩过程,对认识蛇纹岩泥火山渗漏流体的演化至关重要,但关于碳酸盐岩烟囱的形成机制及生长模式仍没有明确认识,影响示踪渗漏流体活动等深入研究。因此,本文选取Quaker蛇纹岩泥火山发育的烟囱状自生碳酸盐岩,依据样品宏观岩石学特征区分出不同生长阶段烟囱,利用体视镜、显微镜和扫描电镜对各类型烟囱进行矿物及其微观形貌观测,结合X射线衍射分析(XRD)、能谱分析及元素含量分析,查明各阶段烟囱的矿物组成及形态特征、矿物生长次序及新生变形规律等,对比新老烟囱以及烟囱横截面内外沉积特征差异,提出烟囱的生长模式,为进一步深刻认识蛇纹岩泥火山流体活动提供基础。
1. 地质背景与样品
马里亚纳弧前是俯冲带体系的一部分,位于马里亚纳海沟以西、马里亚纳火山岛弧和马里亚纳海沟之间的狭长地带(图1a),从南到北长约1 200 km,宽约200~230 km,是西太平洋板块向菲律宾板块俯冲的产物[37]。海相沉积物和增生楔在该俯冲带的弧前体系并不发育,是典型的非增生型俯冲边缘[37-38],由于缺乏增生楔的改造,俯冲板块脱水衍生而成的孔隙流体,能够以最小的变化沿断层运移到海底。深部俯冲板片脱水及蛇纹石化作用导致大量蛇纹岩泥浆裹挟破碎的岩块,在压力与浮力作用下向上运移至海底,在马里亚纳弧前12°~20°N,距海沟轴线30~100 km的狭长范围内,形成串珠状分布的13座大型泥火山[7,39-40]。
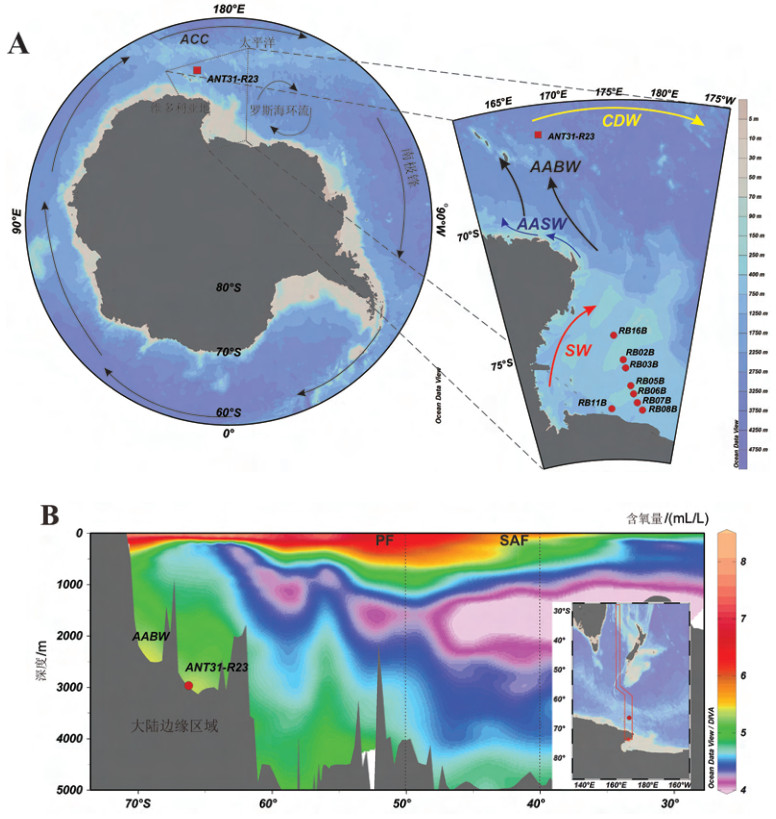
![]() 图 1 马里亚纳弧前蛇纹岩泥火山分布图及泥火山顶海底照片a. 蛇纹岩泥火山分布图,b和c. Quaker蛇纹岩泥火山顶烟囱建造海底照片。图b来自作者与P. Fryer 私人通讯,图c引用自文献 [41]。Figure 1. Distribution map of serpentinite mud volcanoes and photos of the sea bottom at top of the serpentinite mud volcano in the Mariana forearca. distribution map of serpentinite mud volcanoes,b and c are photos of the sea bottom at top of the Quaker seamount showing chimneys. b from private communication with P. Fryer,c after reference [41].
图 1 马里亚纳弧前蛇纹岩泥火山分布图及泥火山顶海底照片a. 蛇纹岩泥火山分布图,b和c. Quaker蛇纹岩泥火山顶烟囱建造海底照片。图b来自作者与P. Fryer 私人通讯,图c引用自文献 [41]。Figure 1. Distribution map of serpentinite mud volcanoes and photos of the sea bottom at top of the serpentinite mud volcano in the Mariana forearca. distribution map of serpentinite mud volcanoes,b and c are photos of the sea bottom at top of the Quaker seamount showing chimneys. b from private communication with P. Fryer,c after reference [41].研究样品来自于马里亚纳弧前的Quaker蛇纹岩泥火山海山(图1a)。样品于2003年3—5月由夏威夷大学P. Fryer教授组织的马里亚纳弧前科考航次由深海无人深潜器(ROV)JasonⅡ在Dive 32和Dive37采集。Quaker蛇纹岩泥火山坐落于马里亚纳北部弧前(图1a),发育于东北-西南走向地垒之上,大致呈南北条形分布,与NE. Quaker和E. Quaker泥火山呈现V字形,高出正常海底约1 000~1 250 m,山顶水深约2 200 m[7]。Quaker海山距离海沟轴线68 km,位于俯冲板块上方21 km。海底声学调查结果显示Quaker海山以蛇纹岩泥浆为主,ROV下潜观测到海底发育显著流体渗漏及大规模烟囱建造(图1b,1c)。
2. 方法
采集的蛇纹岩泥火山自生烟囱使用去离子水清洗,在室温下自然风干。依手标本岩石学特征分类观察并拍照。选取典型部分制成薄片,在OLYMPUS BX53-P光学显微镜下观察其特征,使用高清数码相机拍摄显微图像。样品破裂新鲜断面在OLYMPUS体视镜及扫描电子显微镜COXEM EM-30 PLUS下观测微结构特征,元素成分通过能谱仪OXFROD测定。
挑选的新鲜样品研磨至 200目,用于 X射线衍射(XRD)分析。XRD 分析在中国科学院广州地球化学研究所Rigaku MiniFlex-600型X射线衍射仪上进行,使用Cu靶Kα射线,石墨单色器,测试电压为40 kV,电流为15 mA,扫描角度为3°~80°(2θ),步进扫描,步宽0.02°,狭缝0.1 mm。矿物半定量依面积法确定[42]。
在3个手指状烟囱的横截面用手持牙钻由内向外依次取样,用于主量元素分析。主量元素使用中国科学院地球化学研究所Varian Vista Pro电感耦合等离子体原子发射光谱仪测定,称50 mg粉末样品放入50 mL 离心管中,加入HF和HNO3混合酸消解24 h,然后离心取上清液并转移至干净的特氟龙杯中,在电热板上加热蒸干,最后加入 Rh 内标及稀硝酸上机待测。分析平均标准偏差小于5%,平均相对标准偏差优于5%。
3. 结果
3.1 烟囱类型
依据海底采样过程观测到的流体渗漏情况及烟囱体在海底的风化程度,将Quaker蛇纹岩泥火山海底发育的自生烟囱分为幼年型、成熟型及死亡型烟囱3种类型,三者海底风化程度依次增强。
(1)幼年烟囱:采样观测显示海底流体活动显著,烟囱呈手指状或锥形,手指状直径小于5 cm,烟囱中空,流体通道显著,表皮呈象牙白色或浅黄色,无明显风化特征(图2a,2b)。
![]() 图 2 Quaker海山烟囱样品手标本a. 锥形幼年烟囱底部,外壁浅黄色,烟囱内部呈白色疏松多孔结构,最内侧由白色片状矿物组成,发育中空残余流体通道;b. 手指状幼年烟囱,表皮呈象牙白色,烟囱内部组构与a类似;c. 螺旋手指状成熟烟囱,表皮浅灰色,风化特征明显,烟囱内部由白色疏松组构及最内侧白色片状矿物组成,发育中空残余流体通道;d. 成熟烟囱,烟囱表皮及内部主体与c类似,但中心发育白色片状组构;e. 不规则形态死亡烟囱,表皮棕黑色,风化特征显著,主体由白色致密组构组成,局部发育针状文石可能为残余流体通道充填物。Figure 2. Chimney samples from the Quaker seamounta. an infancy chimney showing light yellow crust, loose and poriferous rim, white flaky core and remnant cavity of fluid channel, b. a finger shaped infancy chimney with bright white crust, but similar inner textures like the sample of a, c. a finger shaped mature chimney showing weathered light gray crust, loose and poriferous rim, white flaky core and remnant cavity of fluid channel, d. a mature chimney with crust and rim texture like the sample of c, but a filled channel, e. a dead chimney with anormal shape showing serious weathered brown crust, with most dense part and trace acicular aragonite. All the scale bars are 3 cm.
图 2 Quaker海山烟囱样品手标本a. 锥形幼年烟囱底部,外壁浅黄色,烟囱内部呈白色疏松多孔结构,最内侧由白色片状矿物组成,发育中空残余流体通道;b. 手指状幼年烟囱,表皮呈象牙白色,烟囱内部组构与a类似;c. 螺旋手指状成熟烟囱,表皮浅灰色,风化特征明显,烟囱内部由白色疏松组构及最内侧白色片状矿物组成,发育中空残余流体通道;d. 成熟烟囱,烟囱表皮及内部主体与c类似,但中心发育白色片状组构;e. 不规则形态死亡烟囱,表皮棕黑色,风化特征显著,主体由白色致密组构组成,局部发育针状文石可能为残余流体通道充填物。Figure 2. Chimney samples from the Quaker seamounta. an infancy chimney showing light yellow crust, loose and poriferous rim, white flaky core and remnant cavity of fluid channel, b. a finger shaped infancy chimney with bright white crust, but similar inner textures like the sample of a, c. a finger shaped mature chimney showing weathered light gray crust, loose and poriferous rim, white flaky core and remnant cavity of fluid channel, d. a mature chimney with crust and rim texture like the sample of c, but a filled channel, e. a dead chimney with anormal shape showing serious weathered brown crust, with most dense part and trace acicular aragonite. All the scale bars are 3 cm.(2)成熟烟囱:采样观测显示有流体活动,烟囱呈螺旋的手指状,直径5~8 cm,烟囱中空,流体通道显著,外壳呈浅灰色,风化现象显著(图2c,2d)。
(3)死亡烟囱:海底采样区域未观察到流体渗漏,烟囱呈不规则形态,外壳棕黑色,风化严重(图2e)。
3.2 矿物组成
对Quaker蛇纹岩泥火山的幼年烟囱内侧、幼年烟囱全岩、成熟烟囱全岩及死亡烟囱进行了XRD物相分析(表1)。自生烟囱矿物组成以碳酸盐矿物为主(52.0%~97.1%)。还发育富镁碱性矿物,主要包括水镁石[Mg(OH)2],水菱镁矿[Mg5(CO3)4(OH)2·4H2O]和水滑石类矿物(水滑石属于阴离子型层状化合物,结构类似水镁石,通用化学式可以表示为[M1-x2+Mx3+(OH)2] [(An-)x/n·mH2O](M为二价或三价金属阳离子,A为阴离子),常见如镁铝碳酸根型水滑石Mg6Al2(OH)16CO3·4H2O。富镁碱性矿物仅在幼年烟囱及成熟烟囱中发育;且幼年烟囱中富镁碱性矿物的种类及丰度均较成熟烟囱更高(表1)。文石仅发育于成熟烟囱及死亡烟囱中,且死亡烟囱的文石含量(33.2%)高于成熟烟囱(23.4%)。所有方解石均为低镁方解石。
表 1 Quaker海山烟囱样品XRD数据Table 1. XRD data of chimneys from Quaker seamount% 样品编号 样品描述 方解石 文石 水菱镁矿 水镁石 水滑石类 石盐 Q1a 幼年烟囱 52.0 0 17.3 18.3 12.3 0 Q1b 幼年烟囱 77.6 0 18.5 3.9 0 0 Q3 成熟全岩 59.8 23.4 0 0 16.8 0 Q4 死亡烟囱 63.9 33.2 0 0 0 2.8 3.3 形貌特征
幼年烟囱、成熟烟囱及死亡烟囱分别具有不同特征的宏观及微观形貌特征。幼年烟囱发育中空的流体通道,通道内壁为薄层白色不规则片状矿物(图3a),向外依次发育白色易碎的疏松多孔组构(图3b),以及坚硬的象牙白色或浅黄色外壁(图2a,2b)。薄片状组构主要由方解石和富镁碱性矿物组成(图4a-f)。方解石呈棱柱状,多具有双晶穿插结构(图4a,4b)。富镁碱性矿物以水镁石为主,还包括水菱镁矿(图4d)和水滑石类矿物(主要是镁铝的层状双金属氢氧化物)(图4e)。水镁石主要呈细叶片状集合体,集合体可构成不规则的放射状结构(图4c),能谱结果显示部分细叶片组构中含少量碳,可能与水镁石溶解或者向水滑石及水菱镁矿转化有关。水菱镁矿呈粗叶片状或者刀刃状(图4d),能谱结果显示由碳、氧和镁元素组成,由于碳含量低于菱镁矿,结合XRD结果应该为水菱镁矿。水滑石类矿物主要是镁铝的层状双金属氢氧化物,呈六方板状,分布于水镁石附近,二者可能为共生或交代关系(图4e)。烟囱外壁和中间的疏松多孔组构主要由方解石组成,富镁碱性矿物组分含量降低,尤其是水镁石含量显著减少,烟囱外壁偶见针状文石交代富镁碱性矿物(图4f)。
![]() 图 3 Quaker海山自生烟囱样品体视镜照片a. 烟囱通道内壁片状富镁碱性矿物,b. 烟囱内壁及外壁之间发育的疏松多孔组构,c. 充填烟囱残余流体通道的针状文石。Figure 3. Stereoscope photos of chimneys from Quaker seamounta. magnesium-rich alkaline mineral from inner rim of an infancy chimney, b. the loose and poriferous texture between crust and inner rim, c. acicular aragonite filling in a channel.
图 3 Quaker海山自生烟囱样品体视镜照片a. 烟囱通道内壁片状富镁碱性矿物,b. 烟囱内壁及外壁之间发育的疏松多孔组构,c. 充填烟囱残余流体通道的针状文石。Figure 3. Stereoscope photos of chimneys from Quaker seamounta. magnesium-rich alkaline mineral from inner rim of an infancy chimney, b. the loose and poriferous texture between crust and inner rim, c. acicular aragonite filling in a channel.![]() 图 4 Quaker蛇纹岩泥火山海底自生烟囱扫描电镜照片a和b幼年烟囱内壁发育的棱柱状方解石和细叶片状水镁石,棱柱状方解石呈双晶或多晶穿插结构;c. 细叶片状水镁石/水菱镁矿集合体呈不规则放射状;d. 粗叶片状或刀刃状水菱镁矿;e. 细叶片状水镁石附近发育六方板状镁铝碳酸根型水滑石类矿物;f. 放射状文石集合体交代水菱镁矿。白色十字表示能谱测量点。Figure 4. SEM images of authigenic chimneys from Quaker serpentinite mud volcanoa & b prismatic calcite with and thin foliated brucite in an inner rim of an infancy chimney, c. aggregations of thin foliated brucite/hydromagnesite showing irregular radial texture. Top right corner is a zoomed-in image, d. thick foliated or blade hydromagnesite, e. hexagonal hydrotalcite occurring near brucite, f. acicular aragonite replacing precursory hydromagnesite. The white cross represent measurement point of EDS.
图 4 Quaker蛇纹岩泥火山海底自生烟囱扫描电镜照片a和b幼年烟囱内壁发育的棱柱状方解石和细叶片状水镁石,棱柱状方解石呈双晶或多晶穿插结构;c. 细叶片状水镁石/水菱镁矿集合体呈不规则放射状;d. 粗叶片状或刀刃状水菱镁矿;e. 细叶片状水镁石附近发育六方板状镁铝碳酸根型水滑石类矿物;f. 放射状文石集合体交代水菱镁矿。白色十字表示能谱测量点。Figure 4. SEM images of authigenic chimneys from Quaker serpentinite mud volcanoa & b prismatic calcite with and thin foliated brucite in an inner rim of an infancy chimney, c. aggregations of thin foliated brucite/hydromagnesite showing irregular radial texture. Top right corner is a zoomed-in image, d. thick foliated or blade hydromagnesite, e. hexagonal hydrotalcite occurring near brucite, f. acicular aragonite replacing precursory hydromagnesite. The white cross represent measurement point of EDS.成熟烟囱内壁也发育类似幼年烟囱的由方解石和富镁碱性矿物组成的薄层片状组构,向外依次发育白色疏松组构及坚硬的浅灰色外壁。显微镜观察显示烟囱内侧由棱柱状方解石和低突起波状消光的矿物组成,该矿物呈不规则扇形,发育微裂纹(图5a,5b),可能为含水矿物失水收缩所致,与扫描电镜下细叶片状水镁石集合体组成的放射状组构类似。烟囱内壁与外壁之间主要发育碳酸盐矿物,主要由棱柱状方解石和交代了不规则扇形富镁碱性矿物的放射状文石组成(图5c,5d)。烟囱外侧主要由方解石和文石组成,方解石多已重结晶呈半自形或他形,充填孔隙的组分为隐晶质或非晶质组分,正交偏光下全消光,可能为报道的无定形镁硅酸盐[36]。成熟烟囱的孔隙度显示从烟囱内侧向外逐渐降低的特征(如图5a,5c,5e的孔隙度依次降低)。
![]() 图 5 Quaker海山烟囱样品显微结构照片a和b 成熟烟囱靠近内壁的部分,孔隙度高,由方解石及呈波状消光的富镁碱性矿物组成,少量文石为交代富镁矿物产物(a单偏光;b正交偏光);c和d成熟烟囱中间部分,孔隙度中等,主要由方解石和文石组成,方解石呈自形-半自形,文石呈放射状交代先驱富镁矿物(c单偏光;d正交偏光);e. 成熟烟囱外壁,低孔隙度,大部分孔隙被充填,几乎全部由方解石和文石组成,方解石重结晶呈半自形-他形(单偏光);f. 充填死亡烟囱残余流体通道的针状文石(正交偏光)。Figure 5. Microstructures of chimneys from Quaker seamounta & b a part near inner rim of a mature chimney with high porosity composed of calcite and magnesium-rich alkaline mineral with wavy extinction. Trace magnesium-rich alkaline mineral were replaced by aragonite(a plane-polarized light; b polarized light), c & d a part between inner rim and outer rim with medium porosity mainly composed of euhedral-subhedral calcite and radial aragonite. Aragonite replaced the precursory magnesium-rich mineral(c plane-polarized light; d polarized light), e. the crust of a mature chimney with low porosity mainly composed of calcite and aragonite. Calcite suffered recrystallization is subhedral-anhedral(plane-polarized light), f. acicular aragonite filling in a channel of a dead chimney(polarized light).
图 5 Quaker海山烟囱样品显微结构照片a和b 成熟烟囱靠近内壁的部分,孔隙度高,由方解石及呈波状消光的富镁碱性矿物组成,少量文石为交代富镁矿物产物(a单偏光;b正交偏光);c和d成熟烟囱中间部分,孔隙度中等,主要由方解石和文石组成,方解石呈自形-半自形,文石呈放射状交代先驱富镁矿物(c单偏光;d正交偏光);e. 成熟烟囱外壁,低孔隙度,大部分孔隙被充填,几乎全部由方解石和文石组成,方解石重结晶呈半自形-他形(单偏光);f. 充填死亡烟囱残余流体通道的针状文石(正交偏光)。Figure 5. Microstructures of chimneys from Quaker seamounta & b a part near inner rim of a mature chimney with high porosity composed of calcite and magnesium-rich alkaline mineral with wavy extinction. Trace magnesium-rich alkaline mineral were replaced by aragonite(a plane-polarized light; b polarized light), c & d a part between inner rim and outer rim with medium porosity mainly composed of euhedral-subhedral calcite and radial aragonite. Aragonite replaced the precursory magnesium-rich mineral(c plane-polarized light; d polarized light), e. the crust of a mature chimney with low porosity mainly composed of calcite and aragonite. Calcite suffered recrystallization is subhedral-anhedral(plane-polarized light), f. acicular aragonite filling in a channel of a dead chimney(polarized light).死亡烟囱主要由方解石和文石组成。烟囱主体部分与成熟烟囱中间及外侧组构类似,由半自形到他形的方解石和文石组成。此外死亡烟囱的显著特征是残余的流体通道被针状文石充填(图3c,5f)。
3.4 主量元素
自生烟囱主量元素含量详见表2。结果显示,自生烟囱主量元素以MgO(1.5%~37.5 %)和CaO(12.2%~53.3 %)为主,Na2O(1.2%~2.9 %)和Al2O3(0.01%~4.01 %)少量。MgO和CaO呈显著负相关关系,且二者含量显示了与烟囱类型及在横截面中位置的良好相关性,即幼年烟囱的MgO高于成熟烟囱,CaO低于成熟烟囱;同一烟囱截面内侧样品的MgO含量高于外侧样品,而外侧样品CaO高于内侧样品(图6)。
表 2 Quaker海山烟囱状自生沉积主量元素分析结果Table 2. The major elemental compositions of the chimneys from Quaker seamount% 样品编号 样品描述 CaO MgO Na2O Al2O3 P2O5 K2O Fe2O3-T Q101 幼年烟囱截面 12.2 37.5 2.0 0.12 0.02 0.05 0.0007 Q102 幼年烟囱截面 21.7 29.0 1.6 0.05 0.03 0.03 0.0015 Q103 幼年烟囱截面 32.1 18.5 1.4 0.02 0.04 0.02 0.0135 Q201 成熟烟囱顶截面 18.6 23.6 2.9 4.01 0.14 0.10 0.0029 Q202 成熟烟囱顶截面 34.2 15.0 1.3 0.21 0.03 0.03 0.0267 Q203 成熟烟囱顶截面 36.5 13.0 1.3 0.03 0.03 0.03 0.0174 Q204 成熟烟囱底截面 28.4 14.8 2.8 2.79 0.09 0.09 0.0020 Q205 成熟烟囱底截面 51.9 2.0 1.2 0.29 0.04 0.01 0.0029 Q206 成熟烟囱底截面 53.3 1.7 1.2 0.04 0.05 0.01 0.0028 Q207 成熟烟囱底截面 52.7 1.5 1.3 0.01 0.05 0.01 0.0019 注:Fe2O3-T代表全岩铁含量。 ![]() 图 6 Quaker海山自生烟囱CaO和MgO元素含量特征图a. CaO和MgO元素含量相关图,b. 烟囱横截面由中心向外的 CaO含量变化图,c. 烟囱横截面由中心向外的 MgO含量变化图。b和c中点位表示微区取样位置,1,2,3,4依次向外。Figure 6. The major elemental compositions of chimneys from Quaker seamounta. CaO vs. MgO plot, b. the CaO contents of micro-drilled samples of chimney cross sections from inner to outer, c. the MgO contents of micro-drilled samples of chimney cross sections from inner to outer. The Arabic numerals represent sampling point in the cross sections. 1, 2, 3, 4 outward successively.
图 6 Quaker海山自生烟囱CaO和MgO元素含量特征图a. CaO和MgO元素含量相关图,b. 烟囱横截面由中心向外的 CaO含量变化图,c. 烟囱横截面由中心向外的 MgO含量变化图。b和c中点位表示微区取样位置,1,2,3,4依次向外。Figure 6. The major elemental compositions of chimneys from Quaker seamounta. CaO vs. MgO plot, b. the CaO contents of micro-drilled samples of chimney cross sections from inner to outer, c. the MgO contents of micro-drilled samples of chimney cross sections from inner to outer. The Arabic numerals represent sampling point in the cross sections. 1, 2, 3, 4 outward successively.4. 讨论
4.1 矿物沉淀及演化
Quaker蛇纹岩泥火山海底发育的自生烟囱由以水镁石为主的富镁碱性矿物和碳酸盐矿物组成。水镁石是海洋蛇纹石化环境流体渗漏活动典型的自生矿物,大西洋中脊Lost City低温热液场及马里亚纳弧前南部海底蛇纹岩出露区海底的流体渗漏活动区(Shinkai Seep Field)均发育水镁石烟囱[8,31,43-45]。蛇纹石化导致流体碱性增强,并通常具有贫镁的特征[3,8,28,34,46],当高pH的碱性渗漏流体在海底与海水混合时,由渗漏流体供给OH−,从海水中获取Mg2+,导致水镁石矿物沉淀[43,45]。但是,水镁石仅在低CO2浓度流体中稳定[47],暴露于海水将导致水镁石变得不稳定并发生溶解。这也是已发现的海底蛇纹石化流体渗漏环境自生烟囱沉积在演化晚期均不含水镁石的原因[35]。
Quaker蛇纹岩泥火山渗漏流体的pH值为9.2,极度亏损镁(Mg2+ 浓度0.5 mmol/kg)[34],与海水混合后有利于水镁石沉淀,幼年烟囱内层水镁石含量最高,说明水镁石为渗漏流体导致的原生沉淀矿物。Quaker蛇纹岩泥火山的死亡烟囱中完全不发育水镁石,指示水镁石在海水环境中发生了溶解作用。Quaker海山自生烟囱的MgO与CaO显著负相关,相关系数R2达0.93(图6a),说明二者之间可能存在转化关系。MgO含量在幼年烟囱、成熟烟囱及死亡烟囱中依次降低,而CaO情况则相反,在幼年烟囱、成熟烟囱及死亡烟囱中依次升高,说明成岩过程中发育水镁石向碳酸盐矿物的转变(公式1和公式2)。Okumura等认为,水镁石溶解的微环境非常富集镁,阻碍方解石沉淀,最终导致碳酸盐以文石相交代水镁石[45]。Quaker海山自生烟囱可见文石交代富镁碱性矿物的特征(图4f,5c,5d),并且XRD结果显示文石在幼年烟囱中不发育,但在成熟烟囱及死亡烟囱中逐渐增多(表1),进一步说明文石形成于自生烟囱在海水环境的成岩过程中,并非流体渗漏活动直接的产物。充填死亡烟囱残余流体通道的针状文石可能直接从海水中沉淀。Quaker蛇纹岩泥火山自生烟囱中文石含量可以指示烟囱的生长阶段和成熟程度,如文石是否发育作为界定成熟烟囱与幼年烟囱的标准,文石含量相对高低定性判断烟囱相对成熟程度。
$$ {\rm{Mg}}{\left( {{\rm{OH}}} \right)_{\rm{2}}} \to {\rm{M}}{{\rm{g}}^{{\rm{2 + }}}}{\rm{ + 2O}}{{\rm{H}}^{\rm{ - }}} $$ (1) $$ {\rm{C}}{{\rm{a}}^{{\rm{2 + }}}}{\rm{ + HC}}{{\rm{O}}_{\rm{3}}}^{\rm{ - }}{\rm{ + O}}{{\rm{H}}^{\rm{ - }}} \to {\rm{CaC}}{{\rm{O}}_{\rm{3}}}{\rm{ + }}{{\rm{H}}_{\rm{2}}}{\rm{O}} $$ (2) 方解石是Quaker蛇纹岩泥火山自生碳酸盐岩的重要矿物组成,在不同类型烟囱中含量变化不大(表1),但在幼年烟囱及成熟烟囱内侧大多呈棱柱状,发育双晶结构,并与水镁石共生产出(图4a,4b),指示来源于渗漏流体导致的沉淀,但在成熟烟囱外壁及死亡烟囱中多呈半自形或他形(图5e),说明方解石在烟囱成岩过程中发生了重结晶作用。前人对马里亚纳弧前另一座蛇纹岩泥火山(Conical海山)自生碳酸盐岩的同位素地球化学研究显示渗漏流体在形成方解石和文石过程中的贡献不同,在形成方解石沉淀流体中的贡献显然大于在形成文石沉淀流体中的贡献[36],说明蛇纹岩泥火山渗漏流体可能更加有利于方解石沉淀,而非文石,这可能与渗漏流体低温、强碱性以及贫镁的特征有关。关于蛇纹岩泥火山与流体渗漏有关的自生碳酸盐矿物的形成,基于渗漏流体富集甲烷,已发现自生烟囱δ13C值最低可达−27.4‰(V-PDB)[48],以及泥火山浅表层蛇纹岩泥的孔隙水中硫酸根与还原性硫的含量负相关[27,49]等现象,推测自生碳酸盐形成过程中存在甲烷缺氧氧化作用(AOM)。但是,尽管自生碳酸盐岩δ13C值分布于−27.4‰~10.2‰,其中大部分仍集中在0附近[13,36,38,48,50],与典型的甲烷缺氧氧化作用形成的自生碳酸盐岩的δ13C值特征(一般低于−30‰)相差甚远[51],且至今未找到有关生物标志物等更加确凿的证据,以致于甲烷缺氧氧化作用在蛇纹岩泥火山自生烟囱形成中是否为普遍现象仍不清楚。此外,本文作者在另外的研究中发现Quaker海山自生烟囱中并未发育碳同位素特别轻的特征,碳酸盐晶格硫(CAS)的硫和氧同位素组成与海水值类似(另文发表),说明并未显示硫酸盐还原作用的改造,说明甲烷缺氧氧化作用对自生方解石形成的贡献可能有限,甲烷缺氧氧化作用对蛇纹岩泥火山自生碳酸盐岩的贡献量可能受来自深部经历高温的蛇纹石化渗漏流体在浅部微生物加入等因素影响[52]。因此,Quaker蛇纹岩泥火山自生烟囱中方解石大多数为高pH,低碳酸盐碱度的渗漏流体与富CO2海水混合的结果。
此外,Quaker蛇纹岩泥火山自生烟囱中还发育水菱镁矿和水滑石类矿物,该二类矿物在陆地蛇绿岩环境流体渗漏导致的自生沉积中有报道过,该环境还发育更多种类的富镁碱性矿物,认为其与环境蒸发以及先驱矿物的富CO2环境蚀变有关[53]。水镁石富CO2的转化,随着CO2分压的增高可能会经历水滑石、纤水菱镁矿、水菱镁矿转变过程[54-55]。如前所述,Quaker蛇纹岩泥火山自生烟囱中先形成的水镁石矿物与渗漏流体隔离后,会最终转化为文石,这是在相对富CO2的海水中新生变形过程。因此,有理由推测水镁石在与渗漏流体隔离不彻底条件下,流体CO2分压相对较低,先转化成了水滑石或水菱镁矿。
总之,Quaker蛇纹岩泥火山渗漏流体具有高pH值,贫镁及低碳酸盐碱度,在喷出海底后与海水混合,形成水镁石和方解石为主的沉淀,水镁石在海水中不稳定,与渗漏流体隔离暴露于海水后经转化为水滑石或水菱镁矿,最终转化为文石,此过程中还伴随方解石的重结晶作用。
4.2 烟囱生长模式
烟囱的生长模式是海底流体渗漏环境、热液和冷泉研究中普遍关心的问题, 目前普遍接受的模式是渗漏流体在浅表层沉积物中或喷出海底后遭遇海水,形成雏形烟囱壁,随着渗漏流体持续不断活动,烟囱发生向上和向内生长[5,56-61]。Quaker蛇纹岩泥火山自生烟囱外侧孔隙度明显小于内侧(图5),外壁风化程度显著大于内侧,说明烟囱外壁经历了更复杂的成岩作用,可能最早形成,支持由外向内生长的模式。从矿物演化规律看,渗漏环境形成的水镁石会最终在海水环境转化为文石,因此水镁石及富镁矿物代表了烟囱新形成组分,新组分元素化学上以高MgO含量为特征;而文石代表成岩改造组分,文石发育说明其先驱的水镁石及富镁矿物较早构成了烟囱体,较老组分元素化学上以低MgO、高CaO含量为特征。对比同一烟囱横截面外侧和内侧的矿物及元素组成,显示外侧样品富镁矿物及MgO含量更低,CaO含量却更高(图6b,6c),且文石发育,支持烟囱外侧先形成,内侧后形成,为由外向内的生长模式。同一烟囱下部横截面样品的CaO含量高于上部横截面,MgO含量低于上部横截面(图6b,6c),说明烟囱下部先形成,支持自下而上的生长。烟囱呈螺旋状,可能与渗漏流体喷出海底后受底流影响改变流体运移方向有关,螺旋状可能说明底流方向多次发生改变。
因此,本文依据Quaker蛇纹岩泥火山自生烟囱的矿物沉淀演化及元素组成特征,提出蛇纹岩自生烟囱由外向内的生长模式。即流体活动初期(幼年期),高pH、贫镁、低碳酸盐碱度的渗漏流体喷出海底后,与海水混合形成水镁石和方解石组成的雏形烟囱外壁,流体持续渗漏过程中,烟囱向内及向上生长,随着内部矿物的沉淀逐渐使雏形烟囱外壁与渗漏流体隔离,导致水镁石受海水影响发生溶解,首先经历向水滑石和水菱镁矿的转变;随着流体继续活动烟囱逐渐发育成熟(成熟期),烟囱体越来越厚,雏形烟囱外壁及烟囱体外侧完全暴露于海水环境,水镁石及所有富镁碱性矿物最终转化为碳酸盐(文石),方解石发生重结晶作用呈半自形或他形,但烟囱最内侧流体渗漏活动仍然维持形成水镁石沉淀;当渗漏流体活动停止(死亡期),烟囱体完全暴露在海水环境,富镁矿物全部转化为碳酸盐,方解石发生重结晶作用,残余流体通道微环境对文石饱和,沉淀交织的针状文石充填物。
5. 结论
(1)依据海底采样信息及烟囱的海底风化程度,将烟囱分为幼年烟囱、成熟烟囱和死亡烟囱。
(2)对比不同类型烟囱、同一烟囱不同位置、以及同一烟囱横截面不同微区取样点的矿物以及元素组成特征,发现渗漏流体有利于水镁石和方解石沉淀,自生烟囱成岩过程中发育水镁石向碳酸盐矿物的转化,伴随MgO的降低及CaO的升高。
(3)在流体活动初期形成幼年烟囱雏形外壁,由方解石和水镁石组成,烟囱由外向内,自下而上生长,随着自生矿物向内生长使外侧矿物逐渐与渗漏流体隔离,暴露于海水中,导致水镁石发生溶解,经历由水菱镁矿及水滑石过渡,最终转化为海水中稳定的文石。
(4)成熟烟囱和死亡烟囱主要由方解石和文石组成。流体渗漏活动停止后,文石充填残余流体通道和孔隙空间,使烟囱孔隙度进一步减小。
致谢:感谢美国夏威夷大学Patty Fryer教授提供样品和采样信息。
致谢: 感谢国家海洋局第一海洋研究所陈志华老师提供的罗斯海表层样,感谢中国科学院海洋所曾志刚研究员以及郭景腾、施江南博士对本文提出的意见与建议。 -
表 1 罗斯海研究区表层沉积物碎屑组分常微量元素比率
Table 1 Major and minor element ratio in detrital components of surface sediments in study area of Ross Sea
站号 Mn/Ti Mo/Ti (×10-4) Ni/Ti (×10-4) Co/Ti (×10-4) Cd/Ti (×10-6) Ca (%) Ti (%) RB02B 0.08 0.84 42.81 22.2 12.65 0.93 0.31 RB03B 0.10 0.63 40.29 20.7 16.36 1.06 0.29 RB05B 0.08 1.24 37.67 19.7 7.94 0.87 0.34 RB06B 0.10 0.56 46.17 23.9 12.25 1.00 0.29 RB07B 0.09 1.35 43.69 23.0 8.39 0.85 0.40 RB08B 0.08 1.54 42.38 22.4 40.75 0.81 0.44 RB11B 0.10 0.71 45.48 23.1 14.06 1.20 0.33 RB16B 0.10 1.06 42.88 21.7 32.24 1.11 0.27 平均值 0.09 0.99 42.67 22.1 18.08 0.98 0.33 标准偏差 0.01 0.36 2.72 1.36 11.91 0.14 0.06 变异系数 7.11% 36.59% 6.38% 6.18% 65.90% 13.95% 17.75% -
[1] Pailler D, Bard E, Rostek F, et al. Burial of redox-sensitive metals and organic matter in the equatorial Indian Ocean linked to precession[J]. Geochimica Et Cosmochimica Acta, 2002, 66(5): 849-865. doi: 10.1016/S0016-7037(01)00817-1
[2] Li C, Love G D, Lyons T W, et al. A stratified redox model for the Ediacaran ocean[J]. Science, 2010, 328(5974): 80-83. doi: 10.1126/science.1182369
[3] Jaccard S L, Galbraith E D, Martinez-Garcia A, et al. Covariation of deep Southern Ocean oxygenation and atmospheric CO2 through the last ice age[J]. Nature, 2016, 530(7589): 207-210. doi: 10.1038/nature16514
[4] Jaccard S L, Galbraith E D. Large climate-driven changes of oceanic oxygen concentrations during the last deglaciation[J]. Nature Geoscience, 2011, 5(2): 151-156. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=ec999cd37b0f8f5720fd8cf4b1d92277
[5] Sigman D M, Hain M P, Haug G H. The polar ocean and glacial cycles in atmospheric CO2 concentration[J]. Nature, 2010, 466(7302): 47-55. doi: 10.1038/nature09149
[6] Anderson R F, Ali S, Bradtmiller L I, et al. Wind-driven upwelling in the Southern Ocean and the deglacial rise in atmospheric CO2[J]. Science, 2009, 323(5920): 1443-1448. doi: 10.1126/science.1167441
[7] Skinner L C, Fallon S, Waelbroeck C, et al. Ventilation of the deep Southern Ocean and deglacial CO2 rise[J]. Science, 2010, 328(5982): 1147-1151. doi: 10.1126/science.1183627
[8] Fischer H, Schmitt J, Lüthi D, et al. The role of Southern Ocean processes in orbital and millennial CO2 variations-A synthesis[J]. Quaternary Science Reviews, 2010, 29(1-2): 193-205. doi: 10.1016/j.quascirev.2009.06.007
[9] Calvert S E. Oceanographic controls on the accumulation of organic matter in marine sediments[J]. Geological Society, London, Special Publications, 1987, 26(1): 137-151. doi: 10.1144/GSL.SP.1987.026.01.08
[10] Calvert S E, Pedersen T F. Anoxia vs. productivity: What controls the formation of organic-carbon-rich sediments and sedimentary rocks?[J]. The American Association of Petroleum Geologists Bulletin, 1990, 74(4): 454.
[11] Galbraith E D, Jaccard S L, Pedersen T F, et al. Carbon dioxide release from the North Pacific abyss during the last deglaciation[J]. Nature, 2007, 449(7164): 890-893. doi: 10.1038/nature06227
[12] Francois R, Altabet M A, Yu E F, et al. Contribution of Southern Ocean surface-water stratification to low atmospheric CO2 concentrations during the last glacial period[J]. Nature, 1997, 389(6654): 929-935. doi: 10.1038/40073
[13] Tribovillard N, Algeo T J, Lyons T, et al. Trace metals as paleoredox and paleoproductivity proxies: An update[J]. Chemical Geology, 2006, 232(1-2): 12-32. doi: 10.1016/j.chemgeo.2006.02.012
[14] Calvert S E, Pedersen T F. Geochemistry of recent oxic and anoxic marine-sediments-implications for the geological record[J]. Marine Geology, 1993, 113(1-2): 67-88. doi: 10.1016/0025-3227(93)90150-T
[15] Brown E T, Le Callonnec L, German C R. Geochemical cycling of redox-sensitive metals in sediments from Lake Malawi: A diagnostic paleotracer for episodic changes in mixing depth[J]. Geochimica Et Cosmochimica Acta, 2000, 64(20): 3515-3523. doi: 10.1016/S0016-7037(00)00460-9
[16] Calvert S E, Pedersen T F. Sedimentary geochemistry of manganese: Implications for the environment of formation of manganiferous black shales[J]. Economic Geology and the Bulletin of the Society of Economic Geologists, 1996, 91(1): 36-47. doi: 10.2113/gsecongeo.91.1.36
[17] Anderson R F, Fleisher M Q, Lehuray A P. Concentration, oxidation-state, and particulate flux of uranium in the Black-Sea[J]. Geochimica Et Cosmochimica Acta, 1989, 53(9): 2215-2224. doi: 10.1016/0016-7037(89)90345-1
[18] Chaillou G, Anschutz P, Lavaux G, et al. The distribution of Mo, U, and Cd in relation to major redox species in muddy sediments of the Bay of Biscay[J]. Marine Chemistry, 2002, 80(1): 41-59. doi: 10.1016/S0304-4203(02)00097-X
[19] Howarth R W, Cole J J. Molybdenum availability, nitrogen limitation, and phytoplankton growth in natural waters[J]. Science, 1985, 229(4714): 653-655. doi: 10.1126/science.229.4714.653
[20] Calvert S E, Pedersen T F. Geochemistry of recent oxic and anoxic sediments: implications for the geological record[J]. Marine Geology, 1993, 113(1-2): 67-88. doi: 10.1016/0025-3227(93)90150-T
[21] Huertadiaz M A, Morse J W. Pyritization of trace-metals in anoxic marine-sediments[J]. Geochimica Et Cosmochimica Acta, 1992, 56(7): 2681-2702. doi: 10.1016/0016-7037(92)90353-K
[22] Erickson B E, Helz G R. Molybdenum(Ⅵ) speciation in sulfidic waters[J]. Geochimica et Cosmochimica Acta, 2000, 64(7): 1149-1158. doi: 10.1016/S0016-7037(99)00423-8
[23] Helz G R, Miller C V, Charnock J M, et al. Mechanism of molybdenum removal from the sea and its concentration in black shales: EXAFS evidence[J]. Geochimica Et Cosmochimica Acta, 1996, 60(19): 3631-3642. doi: 10.1016/0016-7037(96)00195-0
[24] Vorlicek T P, Kahn M D, Kasuya Y, et al. Capture of molybdenum in pyrite-forming sediments: Role of ligand-induced reduction by polysulfides[J]. Geochimica Et Cosmochimica Acta, 2004, 68(3): 547-556. doi: 10.1016/S0016-7037(03)00444-7
[25] Holland M M, Landrum L, Raphael M, et al. Springtime winds drive Ross Sea ice variability and change in the following autumn[J]. Nature Communications, 2017, 8(1): 731. doi: 10.1038/s41467-017-00820-0
[26] Jacobs S S. Bottom water production and its links with the thermohaline circulation[J]. Antarctic Science, 2004, 16(4): 427-437. doi: 10.1017/S095410200400224X
[27] Orsi A H, Johnson G C, Bullister J L. Circulation, mixing, and production of Antarctic Bottom Water[J]. Progress in Oceanography, 1999, 43(1): 55-109. doi: 10.1016/S0079-6611(99)00004-X
[28] Gordon A L, Orsi A H, Muench R, et al. Western Ross Sea continental slope gravity currents[J]. Deep-Sea Research Part Ⅱ-Topical Studies in Oceanography, 2009, 56(13-14): 796-817. doi: 10.1016/j.dsr2.2008.10.037
[29] Orsi A H, Wiederwohl C L. A recount of Ross Sea waters[J]. Deep-Sea Research Part Ⅱ-Topical Studies in Oceanography, 2009, 56(13-14): 778-795. doi: 10.1016/j.dsr2.2008.10.033
[30] Tamura T, Ohshima K I, Nihashi S. Mapping of sea ice production for Antarctic coastal polynyas[J]. Geophysical Research Letters, 2008, 35(7): 284-298. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1029/2007GL032903
[31] Ferrari R, Jansen M F, Adkins J F, et al. Antarctic sea ice control on ocean circulation in present and glacial climates[J]. Proceedings of the National Academy of Sciences of America, 2014, 111(24): 8753-8758. doi: 10.1073/pnas.1323922111
[32] Whitworth T, Orsi A H. Antarctic Bottom Water production and export by tides in the Ross Sea[J]. Geophysical Research Letters, 2006, 33(12): 285-293. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1029/2006GL026357
[33] Rivaro P, Massolo S, Bergamasco A, et al. Chemical evidence of the changes of the Antarctic Bottom Water ventilation in the western Ross Sea between 1997 and 2003[J]. Deep-Sea Research Part I-Oceanographic Research Papers, 2010, 57(5): 639-652. doi: 10.1016/j.dsr.2010.03.005
[34] Van Wijk E M, Rintoul S R. Freshening drives contraction of Antarctic Bottom Water in the Australian Antarctic Basin[J]. Geophysical Research Letters, 2014, 41(5): 1657-1664. doi: 10.1002/2013GL058921
[35] Parker M L, Donnelly J, Torres J J. Invertebrate micronekton and macrozooplankton in the Marguerite Bay region of the Western Antarctic Peninsula[J]. Deep-Sea Research Part Ⅱ-Topical Studies in Oceanography, 2011, 58(13-16): 1580-1598. doi: 10.1016/j.dsr2.2010.08.020
[36] Truesdale G A, Downing A L, Lowden G F. The solubility of oxygen in pure water and sea-water[J]. Journal of Chemical Technology & Biotechnology, 1955, 5(2): 53-62. doi: 10.1002/jctb.5010050201
[37] Merlin O H, Salvador G L, Vitturi L M, et al. Geochemical characteristics of western Ross Sea (Antarctica) sediments[J]. Marine Geology, 1991, 99(1-2): 209-229. doi: 10.1016/0025-3227(91)90092-I
[38] Brumsack H J. Geochemistry of recent toc-rich sediments from the gulf of California and the Black-Sea[J]. Geologische Rundschau, 1989, 78(3): 851-882. doi: 10.1007/BF01829327
[39] Middelburg J J, Delange G J, Vanderweijden C H. Manganese solubility control in marine pore waters[J]. Geochimica Et Cosmochimica Acta, 1987, 51(3): 759-763. doi: 10.1016/0016-7037(87)90086-X
[40] Morford J L, Russell A D, Emerson S. Trace metal evidence for changes in the redox environment associated with the transition from terrigenous clay to diatomaceous sediment, Saanich Inlet, BC[J]. Marine Geology, 2001, 174(1-4): 355-369. doi: 10.1016/S0025-3227(00)00160-2
[41] Algeo T J, Maynard J B. Trace-element behavior and redox facies in core shales of Upper Pennsylvanian Kansas-type cyclothems[J]. Chemical Geology, 2004, 206(3-4): 289-318. doi: 10.1016/j.chemgeo.2003.12.009
[42] Boyle E A. Cadmium: chemical tracer of deepwater paleoceanography[J]. Paleoceanography, 1988, 3(4): 471-489. doi: 10.1029/PA003i004p00471
[43] Rosenthal Y, Lam P, Boyle E A, et al. Authigenic cadmium enrichments in suboxic sediments-precipitation and postdepositional mobility[J]. Earth and Planetary Science Letters, 1995, 132(1-4): 99-111. doi: 10.1016/0012-821X(95)00056-I
[44] Piper D Z, Perkins R B. A modern vs. Permian black shale—the hydrography, primary productivity, and water-column chemistry of deposition[J]. Chemical Geology, 2004, 206(3-4): 177-197. doi: 10.1016/j.chemgeo.2003.12.006
[45] Pujol F, Berner Z, Stüben D. Palaeoenvironmental changes at the Frasnian/Famennian boundary in key European sections: Chemostratigraphic constraints[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 2006, 240(1-2): 120-145. doi: 10.1016/j.palaeo.2006.03.055
[46] Morford J L, Emerson S R, Breckel E J, et al. Diagenesis of oxyanions (V, U, Re, and Mo) in pore waters and sediments from a continental margin[J]. Geochimica et Cosmochimica Acta, 2005, 69(21): 5021-5032. doi: 10.1016/j.gca.2005.05.015
[47] Piper D Z. The metal-oxide fraction of pelagic sediment in the equatorial north Pacific-Ocean-a source of metals in ferromanganese nodules[J]. Geochimica Et Cosmochimica Acta, 1988, 52(8): 2127-2145. doi: 10.1016/0016-7037(88)90193-7
[48] Krishnaswami S. Authigenic transition-elements in Pacific pelagic clays[J]. Geochimica Et Cosmochimica Acta, 1976, 40(4): 425-434. doi: 10.1016/0016-7037(76)90007-7
[49] Grill P B E. The effect of manganese oxide scavenging on molybdenum in saanich inlet, British Columbia[J]. Marine Chemistry, 1974, 2(2): 125-148. doi: 10.1016/0304-4203(74)90033-4
[50] Li Y H. Ultimate removal mechanisms of elements from the ocean[J]. Geochimica Et Cosmochimica Acta, 1981, 45: 1659-1664. doi: 10.1016/0016-7037(81)90001-6
[51] Ceccaroni L, Frank M, Frignani M, et al. Late Quaternary fluctuations of biogenic component fluxes on the continental slope of the Ross Sea, Antarctica[J]. Journal of Marine Systems, 1998, 17(1-4): 515-525. doi: 10.1016/S0924-7963(98)00061-X
[52] Martinson D G. Evolution of the southern ocean winter mixed layer and sea ice: Open ocean deepwater formation and ventilation[J]. Journal of Geophysical Research, 1990, 95(C7): 11641-11654. doi: 10.1029/JC095iC07p11641
[53] Picco P, Bergamasco A, Demicheli L, et al. Large-scale circulation features in the central and western Ross Sea (Antarctica)[J]. Ross Sea Ecology: Springer, 2000: 95-105. doi: 10.1007%2F978-3-642-59607-0_8
[54] Li Y H. Geochemical cycles of elements and human perturbation[J]. Geochimica Et Cosmochimica Acta, 1981, 45(11): 2073-2084. doi: 10.1016/0016-7037(81)90061-2
[55] Ullermann J, Lamy F, Ninnemann U, et al. Pacific-Atlantic Circumpolar Deep Water coupling during the last 500ka[J]. Paleoceanography, 2016, 31(6): 639-650. doi: 10.1002/2016PA002932
[56] Yamamoto A, Abe-Ouchi A, Shigemitsu M, et al. Global deep ocean oxygenation by enhanced ventilation in the Southern Ocean under long-term global warming[J]. Global Biogeochemical Cycles, 2015, 29(10): 1801-1815. doi: 10.1002/2015GB005181
[57] Galbraith E D, Jaccard S L. Deglacial weakening of the oceanic soft tissue pump: global constraints from sedimentary nitrogen isotopes and oxygenation proxies[J]. Quaternary Science Reviews, 2015, 109: 38-48. doi: 10.1016/j.quascirev.2014.11.012
[58] Xiao W S, Esper O, Gersonde R. Last Glacial - Holocene climate variability in the Atlantic sector of the Southern Ocean[J]. Quaternary Science Reviews, 2016, 135: 115-137. doi: 10.1016/j.quascirev.2016.01.023
[59] Wagner M, Hendy I L. Trace metal evidence for a poorly ventilated glacial Southern Ocean[J]. Quaternary Science Reviews, 2017, 170(2): 109-120. https://www.sciencedirect.com/science/article/abs/pii/S0277379117301427
[60] Toggweiler J R. Variation of atmospheric CO2 by ventilation of the ocean's deepest water[J]. Paleoceanography, 1999, 14(5): 571-588. doi: 10.1029/1999PA900033
[61] Wu L, Wang R, Xiao W, et al. Productivity-climate coupling recorded in Pleistocene sediments off Prydz Bay (East Antarctica)[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 2017, 485: 260-270. doi: 10.1016/j.palaeo.2017.06.018
[62] Mcmillan D G, Constable C G, Parker R L. Assessing the dipolar signal in stacked paleointensity records using a statistical error model and geodynamo simulations[J]. Physics of the Earth and Planetary Interiors, 2004, 145(1-4): 37-54. doi: 10.1016/j.pepi.2004.02.011
[63] Prokopenko A A, Khursevich G K. Plio-Pleistocene transition in the continental record from Lake Baikal: Diatom biostratigraphy and age model[J]. Quaternary International, 2010, 219(1-2): 26-36. doi: 10.1016/j.quaint.2009.09.027
-
期刊类型引用(3)
1. 许晓燕,窦衍光,张勇,陈晓辉,孙治雷,白凤龙,翟滨,王利波,邹亮. 冲绳海槽中南部柱状沉积物氧化还原敏感元素赋存机理与环境指示意义. 海洋学报. 2022(10): 63-79 .  百度学术
百度学术
2. 王飞,丛柏林,张朝晖,杨黄浩. 南极罗斯海沉积物中可培养菌株的分离及胞外水解酶活性检测. 极地研究. 2020(01): 68-81 .  百度学术
百度学术
3. XIU Chun,DU Ming,ZHANG Xu,HUO Suxia. Changes of marine productivity and sedimentary environment recorded by biogenic components in the Antarctica Ross Sea since the last deglaciation. Journal of Oceanology and Limnology. 2020(06): 1746-1754 .  必应学术
必应学术
其他类型引用(2)




 下载:
下载: